|
With this tool, we could not only directly demonstrate that agonist-dependent changes in mitochondrial Ca 2+ concentration correlate with an enhancement in mitochondrial ATP concentration, provided that oxidizable substrates are available, but also identify a phenomenon of long-term memory, allowing a prolonged metabolic “priming” that lasts longer than the mitochondrial increase. We thus constructed a chimeric cDNA, which allows the selective targeting of luciferase to the mitochondrial matrix. Indeed, in the cellular environment and in the presence of luciferin, luciferase light emission is a linear function of in a concentration range between 10 −3 and 10 −2 M, i.e., in the physiological range ( 20, 21). The basis for this approach to the measurement of this key cellular parameter was the observation that the affinity of luciferase for ATP, which in vitro is in the micromolar range, is drastically lower in vivo, presumably because of the presence of competing proteins and anions ( 19). For this purpose, we have utilized a targeted recombinant Ca 2+ probe (mitochondrial aequorin, mtAEQ) ( 2) and constructed a tool, a specifically targeted chimera of the ATP-sensitive photoprotein luciferase, with the aim of dynamically monitoring the ATP concentration in the mitochondrial matrix ( m). In this paper, we directly addressed these issues by investigating, in intact living cells, the effect of cytosolic and mitochondrial Ca 2+ signaling on intramitochondrial ATP concentration. The final outcome of this Ca 2+ transfer is expected to be the enhancement of mitochondrial ATP production to balance the increased ATP demand of a stimulated cell ( 15– 18). Recently, it has been shown that, despite the low affinity of the mitochondrial Ca 2+ uptake systems, large increases in matrix parallel the cytosolic Ca 2+ signals, thanks to the close contact between mitochondria and the intracellular Ca 2+ stores ( 14). 
Indeed, three dehydrogenases of the Krebs cycle (pyruvate, isocitrate, and α-ketoglutarate dehydrogenase) are modulated by in the micromolar range ( 12, 13). In this crosstalk, mitochondrial Ca 2+ homeostasis, a process that has attracted a large interest in the past few years ( 2– 11), appears to play a major role. Therefore, oxidative phosphorylation is a complex process regulated at different levels by the interactions of mitochondrial and cytosolic metabolism ( 1). The supply of reduced cofactors (NADH, FADH 2) is ensured by mitochondrial oxidation of substrates derived from glucose, fatty acids, and amino acids via different metabolic pathways. The latter process couples the oxidation of reduced cofactors via the respiratory chain to ATP synthesis by mitochondrial ATP synthase. Most intracellular ATP is derived from cytosolic glycolysis and mitochondrial oxidative phosphorylation. 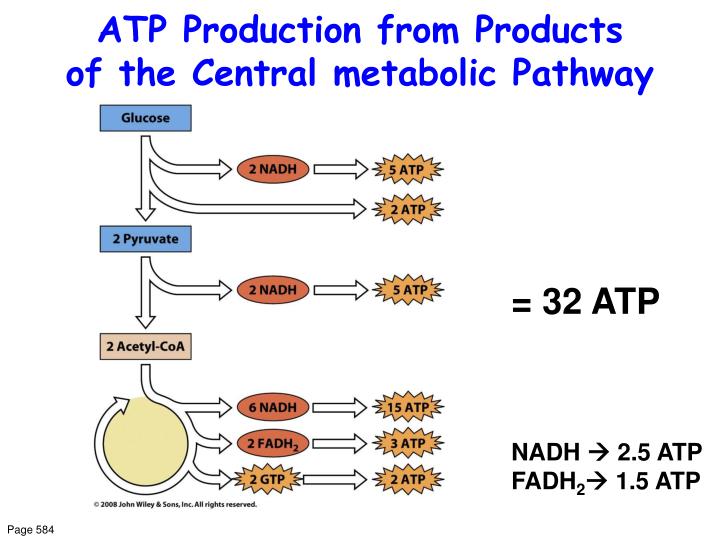
These results demonstrate a direct role of mitochondrial Ca 2+ in driving ATP production and unravel a form of cellular memory that allows a prolonged metabolic activation in stimulated cells.ĪTP is the primary utilizable source of high-energy phosphate bonds within the cell and acts as an allosteric effector of numerous cell processes. Moreover, the Ca 2+ elevation induced a long-lasting priming that persisted long after agonist washout and caused a major increase in m upon addition of oxidative substrates. 
In both HeLa cells and primary cultures of skeletal myotubes, stimulation with agonists evoking cytosolic and mitochondrial Ca 2+ signals caused increases in m and c that depended on two parameters: ( i) the amplitude of the Ca 2+ rise in the mitochondrial matrix, and ( ii) the availability of mitochondrial substrates. Here, we analyzed the significance of Ca 2+ signals for the modulation of organelle function by directly measuring mitochondrial and cytosolic ATP levels ( m and c, respectively) with specifically targeted chimeras of the ATP-dependent photoprotein luciferase. In recent years, mitochondria have emerged as important targets of agonist-dependent increases in cytosolic Ca 2+ concentration.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
